
Zeta-Meter - Model 4.0 - Electrophoretic Mobility and Zeta Potential
Our Zeta-Meter System 4.0 measures electrophoretic mobility and zeta potential. The basic instrument is for aqueous suspending liquids but accessories are available for working with organic suspending liquids.
-
Most popular related searches
Details
Zeta potential measurements are made using a technique called microelectrophoresis. The measurement is very direct. First, your sample is placed in a viewing chamber called an electrophoresis cell. Then an electric field is activated. This causes the colloids to move with a velocity that is proportional to their zeta potential, and in a direction that indicates whether their charge is positive or negative.
Colloids are comfortably observed and tracked using video optics or a high quality stereo microscope. Once your sample is in the cell, you get your first results in a matter of seconds, and complete results in minutes.
Colloids are comfortably observed and tracked usin...
Colloids are comfortably observed and tracked using video optics or a high quality stereo microscope. Once your sample is in the cell, you get your first results in a matter of seconds, and complete results in minutes.
Just fill the cell with your sample, insert the electrodes and connect them to the Zeta-Meter 4.0 unit. It determines the specific conductance of your sample and helps select the appropriate voltage. Energize the electrodes and watch the colloids as they move across a grid in your microscope or video display.
Track one by simply pressing a keypad button and holding it down while your colloid moves across the grid. When you release the button, your colloid’s zeta potential (or electrophoretic mobility) is instantly displayed.
The data display shows you: how many colloids you ...
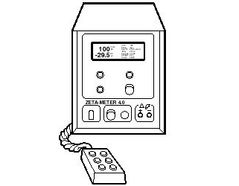
The data display shows you: how many colloids you have tracked, their average zeta potential (or electrophoretic mobility) and the statistical standard deviation of your values. In addition, temperature and average specific conductance are also displayed. You can also review your data in detail at any time by using the keypad to step through each individual tracking value.
We supply software that lets you feed your data directly into the serial port of a personal computer. Our software imports your data into a database that lets you store, search, graph, print and review your data.
The Basics
Understanding zeta potential can help you to understand and control colloidal suspensions. Examples include complex biological systems such as blood, and functional ones like paint. Water, milk, wine, clay, inks, dyes, paper, and pharmaceuticals are good examples of useful colloidal systems. In many cases, the performance of a suspension can be improved by understanding the effects of colloidal behavior on such properties as viscosity, settling, and effective particle size.
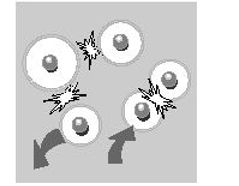
Each colloid carries a 'like' electrical charge which produces a force of mutual electrostatic repulsion between adjacent particles. If the charge is high enough, the colloids will remain discrete, dispersed, and in suspension. Reducing or eliminating the charge has the opposite effect - the colloids will steadily agglomerate and settle out of suspension or form an interconnected matrix.
We can often tailor the characteristics of a suspe...
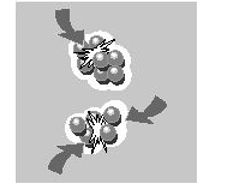
We can often tailor the characteristics of a suspension by understanding how individual colloids interact with one another. At times we may want to maximize the repulsive forces between them in order to keep each particle discrete and prevent them from gathering into larger, faster settling agglomerates. Examples include pharmaceuticals and pastes. Sometimes we have the opposite goal and want to separate the colloids from the liquid. Removing the repulsive forces allows them to form large flocs that settle fast and filter easily. Viscosity is another property that can be modified by varying the balance between repulsion and attraction.
Each charged particle is surrounded by a diffuse layer of neutralizing ions. This causes an electrical potential on the order of millivolts to develop across the diffuse layer between the colloid and the bulk of the liquid. The magnitude of this potential is approximately proportional to the surface charge on the colloid, and it is called the zeta potential. As a result, zeta potential an important measure of the electrokinetic forces involved.
Customer reviews
No reviews were found for Zeta-Meter - Model 4.0 - Electrophoretic Mobility and Zeta Potential. Be the first to review!


